Some specimens may be very tiny; on the order of less than 0.1 cm. Some methods employ the use of mesh cassettes, “tea bag” biopsy pouches, sponges, wrapping paper, etc. in order to contain the specimen and prevent it from escaping the tissue processing cassette. A disadvantage of the above methods is that upon embedding, the specimen has to be handled yet again, possibly resulting in additional fragmentation of the tissue, or possibly complete tissue loss.
A proper method of using wrapping papers is described by Dr. Izak B Dimenstein in his Technical Note published in the Journal of Histotechnology (2016. Vol. 39, No. 3, pages 76-80). This method works very well for fragile tissues such as prostate and breast needle biopsies. Dr. Dimenstein notes that use of sponges/polyester pads may result in a ‘compression artefact”, which can occur during processing. Specifically, tissue needle biopsies may compress and narrow comparable to the size of the pad mesh holes. One solution is to lay out and wrap the core in lens paper, and then sandwich the wrapped specimen between two sponges, which have been pre-soaked in formalin.
With regard to core fragments remaining in the specimen bottle, Dr. Dimenstein recommends filtration through a porous paper, such as the internal layer of a Kimberly-Clark protective mask. This is more reliable than trying to remove fragments with a pipette, where mucous or tiny fragments may stick to the inside of the pipette. He does not recommend filtration through nylon mesh bags, as the tiny fragments are difficult to retrieve during embedding.
With regard to embedding, Dr. Dimenstein recommends the use of a tamper to flatten the core to keep it parallel to the block face. If two cores are present, they should be embedded parallel to each other, and to the horizontal axis of the block. Filtration specimens should be embedded in the manner similar to embedding any aggregate of tissue, carefully grouping the fragments into the middle of the mold.
In addition to the above method, Sakura Finetek has a new product called “embedding gel” that can be used in conjunction with their Paraform cassettes. This material can be used to hold small specimens in place, in proper orientation.
An alternative method is to use a product called “HistoGel”. HistoGel is a liquid at 55 C, and has a gelatin consistency at room temperature. The idea is simply to surround the tiny tissue fragment(s) with liquefied HistoGel, allow it to cool to room temperature (two minutes), thereby trapping the tissue fragment in the HistoGel, much the same way fruit is embedded in the gelatin of a Jell-O fruit mold. The resulting “button” of HistoGel containing the tissue is placed into a tissue processing cassette and processed as usual. (Note: do not use microwave processing.) During embedding, the button is simply removed from the cassette and embedded as usual. Additional slides may have to be taken in order to reach the fragment; however, the tissue cannot be lost in processing. This fact far outweighs the extra effort involved during the surgical grossing procedure.
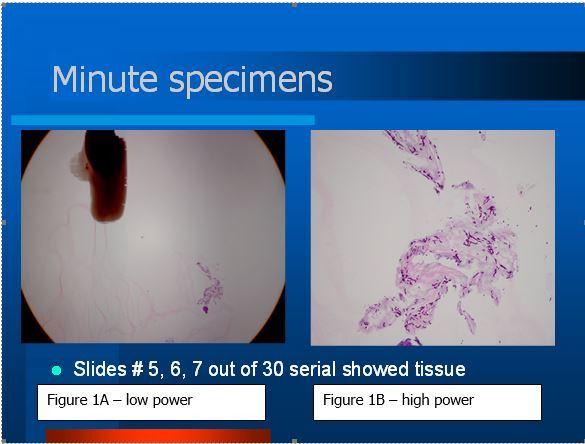
Figure 1A shows a black ink dot, marking the presence of a small group of cells that represent a tiny fragment submitted in Histogel. Figure 1B is a high power micrograph of the specimen stained with PAS. Fungi are clearly demonstrated. The specimen was so small that it had to be serially sectioned. Slides #5, 6, and 7 out of 30 slides showed the tissue fragment.
REFERENCES:
- Theory and Practice of Histological Techniques. JD Bancroft, A Stevens ed. Churchill Livingstone, NY. Fourth edition. 1996
- Theory and Practice of Histotechnology. DC Sheehan, BB Hrapchak. CV Mosby Company, St. Louis. First edition. 1980.
- Luna L. AFIP. Manual of Histologic Staining Methods. Third Edition. McGraw-Hill. p39. 1968. As modified by CM Chapman
- Dermatopathology Laboratory Techniques. CM Chapman, I Dimenstein. In press.


